The drug was approved by the U.S. Food and Drug Administration (FDA) on April 28th, 2005. On 2nd November 2009, it was approved for the expanded use as a First-line treatment for Type 2 Diabetes. On 20th October 2011, it was approved for use with Insulin Glargine in the U.S.
In January 2012, Bydureon, a once-weekly injection was finally approved by the FDA, which was rejected twice earlier in 2010 because of heart rhythm abnormalities. (Bydureon is a longer-lasting version of Byetta.)
When Byetta was introduced in the year 2005, it was unique and different from other drugs. Hundred of patients who used it for Type-2 diabetes till now have experienced pancreatitis side effects or been diagnosed with pancreatic cancer. Some of them even have died due to the complication related to this drug. Unknown side effects of Byetta have been hidden from the public, which resulted in the filing of lawsuits against the manufacturer of this drug. Approximately 1000 cases against Byetta are currently pending trials in a federal court in California.
In the year 2005, makers of Byetta, Amylin Pharmaceuticals, and Eli Lilly and Company announced that the drug for controlling blood sugar in Type 2 diabetes was approved by FDA.
During the press release in the year 2005, the CEO of Eli Lilly and Company stated that Byetta has special content and design which can provide a new option for Type 2 diabetes patients and the launch of the drug in the market is a big milestone in itself.
Bristol-Myers Squibb and AstraZeneca were tempted to acquire Amylin Pharmaceuticals for around $7 B. AstraZeneca later purchased the diabetes division of Bristol-Myers Squibb’s that includes Byetta for more than $4 billion. By the year 2012, Byetta earned billion by its sale. Physicians and patients found this drug at that time a new weapon to fight the diabetes epidemic.
As per lawsuits filed, plaintiffs claim that manufacturers of Byetta did not perform the safety test to check the side effects of the drug, before seeking approval for its sale. Later on when makers of Byetta came to know about the shortcoming of their drug. They failed to warn about the risk to doctors as well as patients.
The German Medical Association in its study has stated that a higher rate of pancreatic cancer was observed in patients intaking Byetta. A clear linkage was shown between exenatide, an active ingredient in Byetta with the development of pancreatic cancer.
By the year 2013, dozens of pancreatic cancer lawsuits against manufacturers of Byetta were filed together in the Southern District of California as multidistrict litigation (MDL). Within two years the lawsuits in the MDL swelled into hundreds. The plaintiffs have various allegations like Byetta manufacturers did not mention the risk in their warning label, the defective design are unreasonably dangerous and most importantly the makers of this Type-2 diabetes drug allegedly kept the fact about the risk of pancreatic cancer development hidden from the public.
Serious Alleged Injuries May Include:
- Pancreatic Cancer
- Kidney Failure
- Severe Allergic Reaction
- Pancreatitis
- Pre-Cancerous Cellular Changes Called Pancreatic Duct Metaplasia
- Hemorrhagic Pancreatitis
- Necrotizing Pancreatitis
- Thyroid Cancer
FDA Safety Warnings:
- In October 2007, the FDA asked the Amylin Pharmaceuticals, Inc. to include a warning about pancreatitis in precautions section of the product label.
- In August 2008, the FDA issued a prominent warning against Hemorrhagic and Necrotizing Pancreatitis.
- On November 02, 2009, FDA approved revisions in drug label to include information of altered kidney function, acute renal failure, and insufficiency.
- In March 2013, the FDA issued a Safety communication that indicated further reports will be investigated for possible increased risks of pancreatitis and pre-cancerous findings.
- In May 2013, the FDA issued safety communications linking the Byetta and other incretin mimetics to serious complications, such as pancreatic diseases.
What is Byetta?
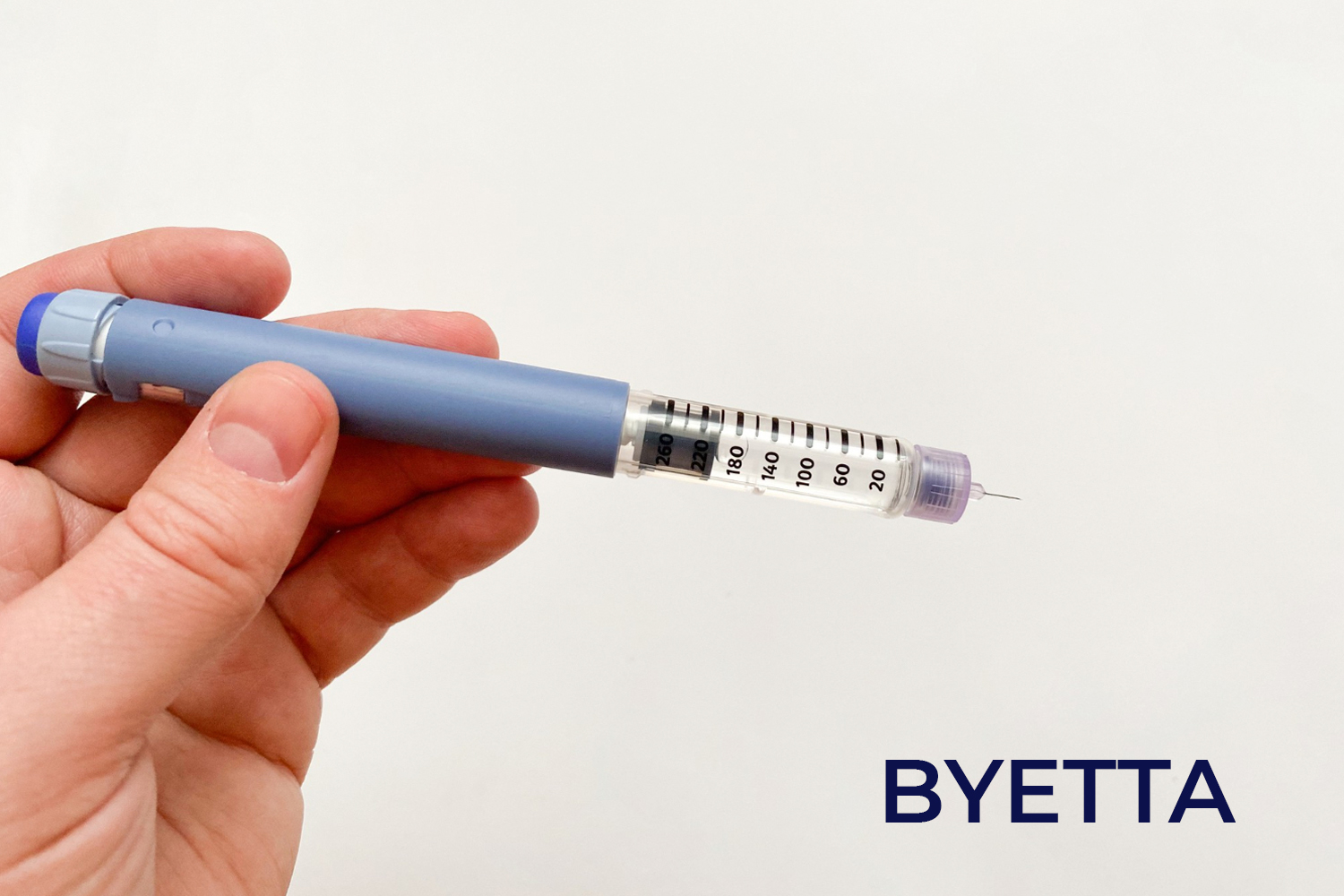
Byetta is used with a diet and exercise program to help lower sugar levels (glucose)in blood in grown-ups with type 2 diabetes. Byetta is not currently approved for use in children below 18 years of age.
Byetta includes exenatide, which is a kind of medicine known as a glucagon-like peptide-1 (GLP-1) agonist. Byetta is given by injection.
It comes in a prefilled injection pen. You use an injection pen to give yourself an injection under your skin (subcutaneous injection). You should take your dose before each of your two main meals of the day (such as breakfast and dinner).
If you are taking Byetta, you may be given other medications such as metformin, sulfonylurea, or both to help lower your blood sugar levels.
Byetta is effective when used alone and in combination with various combinations of diabetes medications. Lower your hemoglobin A1c (HbA1c) levels, which measure your blood sugar level over the past three months.
In one Byetta clinical study used, people receiving Byetta had their HbA1c intermediate levels reduced by 0.7% to 0.9% after 24 weeks. This was compared with a 0.2% reduction in people receiving placebo (treatment without an active drug). People receiving Byetta also had their normal blood sugar reduced by 17 mg / dL to 19 mg / dL, compared to 5 mg / dL in people receiving placebo
Similar results have been observed in other medical studies in which people received Byetta and other anti-diabetic drugs. These include metformin, sulfonylurea (like glipizide), thiazolidinedione (similar to pioglitazone), and insulin glargine.
Byetta is only available as a brand name tree. Currently not available in the normal way.
Byetta contains one active drug ingredient: exenatide. Exenatide is also available in a long-acting form such as Bydureon named after the drug.
Byetta comes as a completed injection pen. It is available in two doses: 5 mcg per dose and 10 mcg per dose. Each pen contains 60 volumes.
Type 2 diabetes
Your first dose of Byetta will probably be injected at 5 mcg twice a day, an hour before your two main meals. Most people give themselves one injection an hour before breakfast and another one an hour before dinner.
However, if you do not eat a lot of breakfast, you may choose to get your first injection an hour before lunch. Your second injection will still be an hour before your meal, as long as these foods are separated for at least six hours. Ask your doctor for advice if you are not sure when to give your injections.
After four weeks, your doctor may increase your dose to 10 mcg twice a day. This will depend on how good your blood sugar levels respond to Byetta injections. Your medical expert will decide which dose is best for you.
Legal Updates:
In 2013, Byetta cases along with several diabetes medications (Victoza, Januvia, and Janumet) were centralized in a multidistrict litigation MDL 2452 (In Re: Incretin Mimetics Products Liability Litigation presided by Judge, Anthony J. Battaglia) in the Southern District of California. Lawsuits filed alleged that though the manufacturers knew about the link of their product to cancer side effects in several studies, the details were not made public. Byetta lawsuits were filed across the nation, claiming that the drug caused the patients to develop thyroid and pancreatic cancer.
In November 2015, Judge Battaglia granted a summary judgment in favor of the defendants, following a preemption protocol dismissing all the cases in the MDL.
In September 2016, plaintiff’s attorneys filed an appeal to the Ninth Circuit U.S. Court of Appeals on behalf of the MDL stating that Judge Battaglia misinterpreted the U.S. Supreme Court preemption law.
In November 2017, the U.S. Court of Appeals for the Ninth Circuit reversed and vacated the Federal District Court Judge’s ruling, thereby reinstating the dismissed federal lawsuits and sending them back to the Federal District Court in California for further proceedings.
Evidence:
- Duration Of Byetta Usage
- Usage Of Byetta In Pharmacy Records
- Indications For Usage Of Byetta In Medical Records
- Complications And Their Treatment After Byetta Intake
Medical Record Review and claim validation of Byetta case should take approximately 3 hours in most instances; however, this approximation may vary in cases based on the volume of records.